[ad_1]
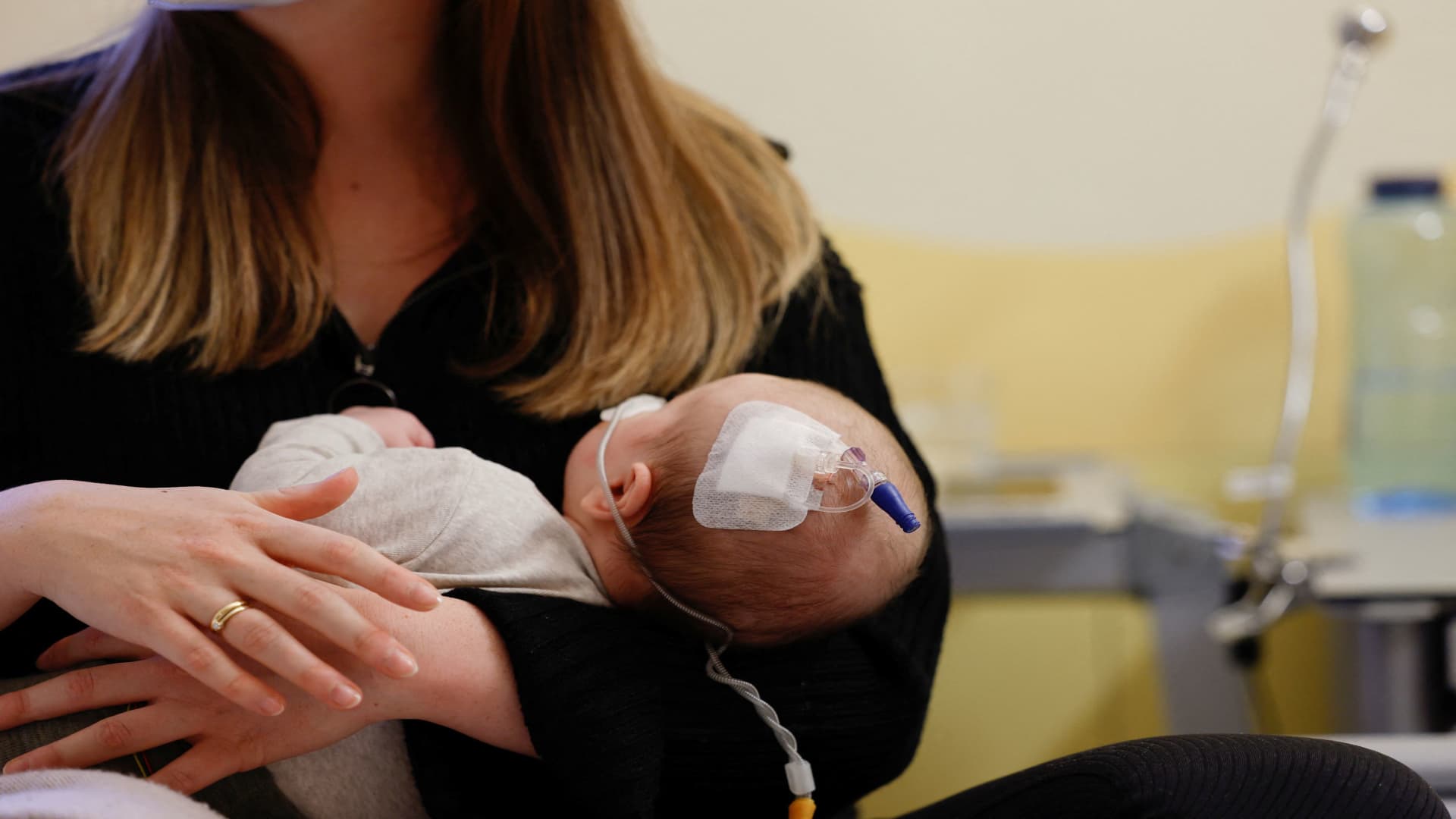
A RSV-infected child receives treatment, as RS-Virus infections spread among children at Missio children’s clinic Moenchberg, in Wuerzburg, Germany, December 2, 2022.
Heiko Becker | Reuters
The Centers for Disease Control and Prevention expedited the release of more than 77,000 additional doses of a new drug designed to protect infants from respiratory syncytial virus amid an ongoing shortage of the treatment in the U.S.
The CDC’s move late Thursday to increase the availability of Beyfortus, a monoclonal antibody from Sanofi and AstraZeneca that won approval in August, comes as RSV cases rise in some parts of the country ahead of the holiday season. The drug is one of two available treatments in the U.S. that can protect infants from the virus, which is the leading cause of hospitalization among babies nationwide.
Sanofi and AstraZeneca did not immediately respond to a request for comment on the CDC’s announcement.
Hospitals and pediatricians have been struggling to stock Beyfortus due to what Sanofi has described as “unprecedented demand” for the treatment. The shortage – and other issues related to insurance coverage – threatens to prevent infants from receiving critical protection against RSV.
RSV is a common respiratory infection that usually causes mild, cold-like symptoms, but can present as more severe cases in children and older adults.
Each year, the virus kills a few hundred children younger than 5, and 6,000 to 10,000 seniors, according to the CDC. RSV also causes around 58,000 to 80,000 hospitalizations among children younger than 5 years old each year, the CDC said.
The CDC said the additional doses will be distributed immediately to physicians and hospitals through commercial channels and the Vaccines for Children Program, which covers the cost of the shots for uninsured and underinsured kids.
CDC said the agency, along with the Food and Drug Administration, will continue to be in close contact with the drug manufacturers to ensure availability of additional doses through the end of this year and early 2024 to meet demand.
“CDC and FDA are committed to expanding access to this important immunization so that more parents have peace of mind during the winter virus season,” said the CDC’s principal deputy director, Dr. Nirav Shah, in a statement.
The U.S. started to see a sharp uptick in RSV cases in the middle of October. Nearly 5,000 cases were detected through testing in the U.S. in the week ended Nov. 4, the highest level since last winter, according to the CDC website.
The U.S. suffered an unusually severe RSV season last year. Cases of the virus in children and older adults overwhelmed hospitals across the country, largely because the public stopped practicing Covid pandemic health measures that had helped to keep the spread of RSV low.
[ad_2]





